The most industrially significant amines are prepared from ammonia by alkylation in amine alkylation. Haloalkanes react with amines to give a corresponding alkyl-substituted amine, with the release of a halogen acid. Such reactions, which are most useful for alkyl iodides and bromides, are rarely employed because the degree of alkylation is difficult to control.
Nitriles are reduced to amines using hydrogen in the presence of a nickel catalyst, although acidic or alkaline conditions should be avoided to avoid hydrolysis of -CN group. LiAlH4 is more commonly employed for the reduction of nitriles on the laboratory scale. Similarly, LiAlH4 reduces amides to amines.
Aniline and its derivatives are prepared by reduction of the nitroaromatics. Many laboratory methods exist for the preparation of amines, many of these methods being rather specialized.
Reaction name  | Substrate  | Comment |
|---|---|---|
| Gabriel synthesis | organohalide | reagent: potassium phthalimide |
| Staudinger reduction | Azide | This reaction also takes place with a reducing agent such as lithium aluminium hydride. |
| Schmidt reaction | carboxylic acid | |
| Aza-Baylis-Hillman reaction | imine | Synthesis of allylic amines |
| Hofmann degradation | amide | This reaction is valid for preparation of primary amines only. Gives good yields of primary amines uncontaminated with other amines. |
| Hofmann Elimination | Quaternary ammonium salt | upon treatment with strong base |
| Amide reduction | amides | |
| Nitrile reduction | nitriles | |
| Reduction of nitro compounds | nitro compounds | can be accomplished with elemental zinc, tin or iron with an acid. |
| Amine alkylation | haloalkane | |
| Delepine reaction | organohalide | reagent hexamine |
| Buchwald-Hartwig reaction | aryl halide | specific for aryl amines |
| Menshutkin reaction | tertiary amine | reaction product a quaternary ammonium cation |
| hydroamination | alkenes and alkynes | |
| Hofmann-Löffler reaction | haloamine |
Reactions
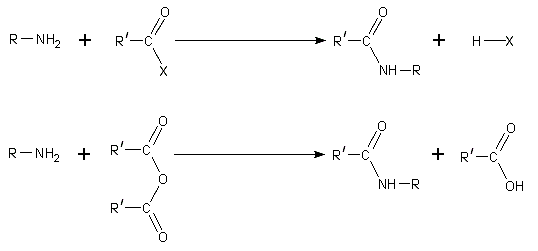
The dominant reactivity of amines is their nucleophilicity. Most primary amines are good ligands for metal ions to give coordination complexes. Amines are alkylated by alkyl halides. Acyl chlorides and acid anhydrides react with primary and secondary amines to form amides (the "Schotten-Baumann reaction").
Similarly, with sulfonyl chlorides, one obtains sulfonamides. This transformation, known as the Hinsberg reaction, is a chemical test for the presence of amines.
Because amines are basic, they neutralize acids to form the corresponding ammonium salts R3NH+. When formed from carboxylic acids and primary and secondary amines, these salts thermally dehydrate to form the corresponding amides.
Amines react with nitrous acid to give diazonium salts. The alkyl diazonium salts spontaneously decompose by losing N2 to produce a mixture of alkenes, alkanols or alkyl halides, with alkanols as the major product. This reaction is of little synthetic importance because the diazonium salt formed is too unstable.

Primary aromatic amines, such as aniline ("phenylamine") form more stable diazonium salts, which can be isolated in the crystalline form. These species undergo a variety of synthetically useful transformations. With cuprous cyanide the corresponding nitrile is formed. Arenediazonium ions undergo coupling with electron-rich aromatic compounds such as a phenol to form an azo compound. These species are widely used as dyes.

Imine formation is an important reaction. Primary amines react with ketones and aldehydes to form imines. In the case of formaldehyde (R' = H), these products typically exist as cyclic trimers.
- RNH2 + R'2C=O → R'2C=NR + H2O
Reduction of the imines gives secondary amines:
- R'2C=NR + H2 → R'2CH-NHR
Similarly, secondary amines react with ketones and aldehydes to form enamines:
- R2NH + R'(R"CH2)C=O → R"CH=C(NR2)R' + H2O
An amine reaction overview is given below:
Reaction name  | Reaction product  | Comment |
|---|---|---|
| Amine alkylation | amines | degree of substitution increases |
| Schotten-Baumann reaction | amides | Reagents: acyl chlorides, acid anhydrides |
| Hinsberg reaction | sulfonamides | Reagents: sulfonyl chlorides |
| Amine-carbonyl condensation | imines | |
| Organic oxidation | nitroso compounds | Reagent: peroxymonosulfuric acid |
| Organic oxidation | diazonium salt | Reagent: nitrous acid |
| Zincke reaction | Zincke aldehyde | reagent pyridinium salts , with primary and secondary amines |
| Emde degradation | tertiary amine | reduction of quaternary ammonium cations |
| Hofmann-Martius rearrangement | aryl substituted anilines | |
| Von Braun reaction | Organocyanamide | By cleavage (tertiary amines only) with cyanogen bromide |
Biological activity
Many kinds of biological activity produce amines by breakdown of amino acids. Many natural neurotransmitters like epinephrine, norepinephrine, dopamine, serotonine, histamine are amines.
Application of amines
Dyes
Primary aromatic amines are used as a starting material for the manufacture of azo dyes. It reacts with nitric(III) acid to form diazonium salt, which can undergo coupling reaction to form azo compound. As azo-compounds are highly coloured, they are widely used in dyeing industries, such as:
- Methyl orange
- Direct brown 138
- Sunset yellow FCF
- Ponceau
Drugs
Many drugs are designed to mimic or to interfere with the action of natural amine neurotransmitters, exemplified by the amine drugs:
- Chlorpheniramine is an antihistamine that helps to relieve allergic disorders due to cold, hay fever, itchy skin, insect bites and stings.
- Chlorpromazine is a tranquillizer that sedates without inducing sleep. It is used to relieve anxiety, excitement, restlessness or even mental disorder.
- Ephedrine and Phenylephrine, as amine hydrochlorides, are used as decongestants.
- Amphetamine, Methamphetamine, and Methcathinone are amines that are listed as controlled substances by the DEA.
- Amitriptyline, Imipramine, Lofepramine and Clomipramine are tricyclic antidepressants and tertiary amines
- Nortriptyline, Desipramine, and Amoxapine are tricyclic antidepressants and secondary amines
- (The tricyclics are grouped by the nature of the final amine group on the side chain.)
Gas treatment
- Aqueous monoethanolamine (MEA), diglycolamine (DGA), diethanolamine (DEA), diisopropanolamine (DIPA) and methyldiethanolamine (MDEA) are widely used industrially for removing carbon dioxide (CO2) and hydrogen sulfide (H2S) from natural gas streams and refinery process streams. They may also be used to remove CO2 from combustion gases / flue gases and may have potential for abatement of greenhouse gases.
Safety
Low molecular weight amines are toxic and some are easily absorbed through the skin. Many higher molecular weight amines are highly active biologically.

No comments:
Post a Comment